|
   |
|
Promising Vaccine Candidates Chemical synthesis of a polypeptide backbone derived from the primary sequence of the cancer protein NY-ESO-1 enabled by kinetically controlled ligation and pseudoprolines. The cancer protein NY-ESO-1 has been shown to be one of the most promising vaccine candidates although little is known about its cellular function. Using a chemical protein strategy, the 180 amino acid polypeptide, tagged with an arginine solubilising tail, was assembled in a convergent manner from four unprotected peptide α- thioester peptide building blocks and one cysteinyl polypeptide, which were in turn prepared by Boc and Fmoc SPPS respectively. To facilitate the assembly by ligation chemistries, non-native cysteines were introduced as chemical handles into the polypeptide fragments; pseudoproline dipeptides and microwave assisted Fmoc SPPS were crucial techniques to prepare the challenging hydrophobic Cterminal fragment. Three sequential kinetically controlled ligations, which exploited the reactivity between peptide arylthioesters and peptide alkylthioesters, were then used in order to assemble the more tractable N-terminal region of NY-ESO-1. The ensuing 147 residue polypeptide thioester then underwent successful final native chemical ligation with the very hydrophobic C-terminal polypeptide bearing an N-terminal cysteine affording the 186 residue polypeptide as an advanced intermediate en route to the native NY-ESO-1 protein. This article is protected by copyright. All rights reserved. © 2015 Wiley Periodicals, Inc. Novel Therapeutics For Alcohol Intoxication and Dependence Oxytocin prevents ethanol actions at {delta} subunit-containing GABAA receptors and attenuates ethanol-induced motor impairment in rats.Bowen MT, Peters ST, Absalom N, Chebib M, Neumann ID, McGregor IS., PNAS, 2015 Feb 23. doi:10.1073/pnas.1416900112 [Epub ahead of print] Even moderate doses of alcohol cause considerable impairment of motor coordination, an effect that substantially involves potentiation of GABAergic activity at δ subunit-containing GABAA receptors (δ-GABAARs). Here, we demonstrate that oxytocin selectively attenuates ethanol-induced motor impairment and ethanol-induced increases in GABAergic activity at δ-GABAARs and that this effect does not involve the oxytocin receptor. Specifically, oxytocin (1 μg i.c.v.) given before ethanol (1.5 g/kg i.p.) attenuated the sedation and ataxia induced by ethanol in the open-field locomotor test, wire-hanging test, and righting-reflex test in male rats. Using two-electrode voltage-clamp electrophysiology in Xenopus oocytes, oxytocin was found to completely block ethanol-enhanced activity at α4β1d and α4β3d recombinant GABAARs. Conversely, ethanol had no effect when applied to α4β1 or α4β3 cells, demonstrating the critical presence of the d subunit in this effect. Oxytocin had no effect on the motor impairment or in vitro effects induced by the δ-selective GABAAR agonist 4,5,6,7-tetrahydroisoxazolo(5,4- c)pyridin-3-ol, which binds at a different site on δ-GABAARs than ethanol. Vasopressin, which is a nonapeptide with substantial structural similarity to oxytocin, did not alter ethanol effects at δ-GABAARs. This pattern of results confirms the specificity of the interaction between oxytocin and ethanol at δ- GABAARs. Finally, our in vitro constructs did not express any oxytocin receptors, meaning that the observed interactions occur directly at δ-GABAARs. The profound and direct interaction observed between oxytocin and ethanol at the behavioral and cellular level may have relevance for the development of novel therapeutics for alcohol intoxication and dependence.
Engineering Potent and Selective Analogs of GpTx-1, a Tarantula Venom Peptide Antagonist of the NaV1.7 Sodium Channel Murray JK, Ligutti J, Liu D, Zou A, Poppe L, Li H, Andrews KL, Moyer BD, McDonough SI, Favreau P, St�cklin R, Miranda LP., J. Med. Chem., Just Accepted Manuscript DOI: 10.1021/jm501765v Publication Date (Web): February 6, 2015 Copyright � 2015 American Chemical Society NaV1.7 is a voltage-gated sodium ion channel implicated by human genetic evidence as a therapeutic target for the treatment of pain. Screening fractionated venom from the tarantula Grammostola porteri led to the identification of a 34- residue peptide, termed GpTx-1, with potent activity on NaV1.7 (IC50 = 10 nM) and promising selectivity against key NaV subtypes (20x and 1000x over NaV1.4 and NaV1.5, respectively). NMR structural analysis of the chemically synthesized three disulfide peptide was consistent with an inhibitory cystine knot motif. Alanine scanning of GpTx-1 revealed that residues Trp29, Lys31, and Phe34 near the C-terminus are critical for potent NaV1.7 antagonist activity. Substitution of Ala for Phe at position 5 conferred three hundred-fold selectivity against NaV1.4. A structure-guided campaign afforded additive improvements in potency and NaV subtype selectivity, culminating in the design of [Ala5,Phe6,Leu26,Arg28]GpTx-1 with a NaV1.7 IC50 value of 1.6 nM and >1000x selectivity against NaV1.4 and NaV1.5. One Step Closer to a Cure Glucagon-like peptide-1 protects the murine hippocampus against stressors via Akt and ERK1/2 signaling. Yoshino Y, Ishisaka M, Tsujii S, Shimazawa M, Hara H., Biochem Biophys Res. Commun. ,2015 Feb 7. doi: 10.1016/j.bbrc.2015.01.098. [Epub ahead of print] Alzheimer's disease (AD) is a common neurodegenerative disease characterized by cognitive dysfunction and neuronal cell death in the hippocampus and cerebral cortex. Glucagonlike peptide-1 (GLP-1) is an insulinotropic peptides. GLP-1- associated medicines are widely used as treatments for type 2 diabetes. In addition, they have been shown to ameliorate pathology in AD mouse models. Here, we investigated the effects of GLP-1 on different stressors in murine hippocampal HT22 cells. GLP-1 (7-36)prevented H2O2-, l-glutamate-, tunicamycin-, thapsigargin-, and amyloid β1-42-induced neuronal cell death in a concentration-dependent manner. GLP-1 (7-36) treatment for 1 h significantly increased phosphorylated Akt and extracellular signal-regulated kinase 1 and 2 (ERK1/2) when compared with vehicle-treatment. These results suggest that GLP-1 (7-36) is protective against these stressors via activation of survival signaling molecules, such as Akt and ERK1/2 in HT22 cells. In conclusion, GLP-1 and activators of the GLP-1 receptor might be useful targets for the treatment of AD. Copyright © 2015 Elsevier Inc. All rights reserved.
Glucagon-Like Peptide-1 (7-36) [GLP-1 (7-36)] has shown potential in the treatment of type II diabetes. For example, it restores myocardial insulin sensitivity and prevents progressive heart failure. (Chen M, et al. Cardiovasc. Diabetol., 2014, 13, 115) GLP-(7-36) also has protective effects in the brain. It has protective effects on brain ischemia/reperfusion damage in diabetic rats. (Zhao L, et al., Brain Res., 2015 Jan. 16. doi: 10.1016/j.brainres.2015.01.014 [Epub. ahead of print]) and protects synaptic and learning functions from neuroinflammation in rodents (Iwai T, et al., J. Neurosci. Res., 2014, 92, 946-54). Administered intrathecally, GLP-1(7-36) suppresses pain hypersensitivity. (Gong N, et al, J. Neurosci, 2014, 34, 5322-34) AAPPTec offers glucagon-like peptide-1 (7-36) for research applications.
4-Cyanophenylalanine 4-Cyanophenylalanine residues may be utilized as non-invasive fluorescent and IR probes. 4-Cyanophenylalanine residues have been used as probes in 2D IR spectroscopy (Urbanek DC, et al., J. Phys. Chem. Lett., 2010, 1, 3311-5; Chung JK, et al., J. Am. Chem. Soc. , 2012, 134, 12118-24). The 4-cyanophenylalanine is easily substituted for a phenylalanine residue in synthetic peptides. Methods of genetically encoding 4-cyanophenylalanine have also been developed ( Schultz KC, et al., J. Am. Chem. Soc., 2006, 128, 13984-5). EJ Petersson and coworkers have utilized 4- cyanophenylalanine with backbone thioamides as FRET pairs (Goldberg JM, et al., J. Am. Chem. Soc., 2010, 132, 14718-20; Wissner, EF, et al., J. Am. Chem. Soc., 2013, 135, 6529-40). AAPPTec offers Boc-protected, Fmoc-protected, or unprotected L-4-cyanophenylalanine for use in structural studies. These products are available in convenient catalog quantities or you may request a bulk quotation by email to sales@aapptec.com.
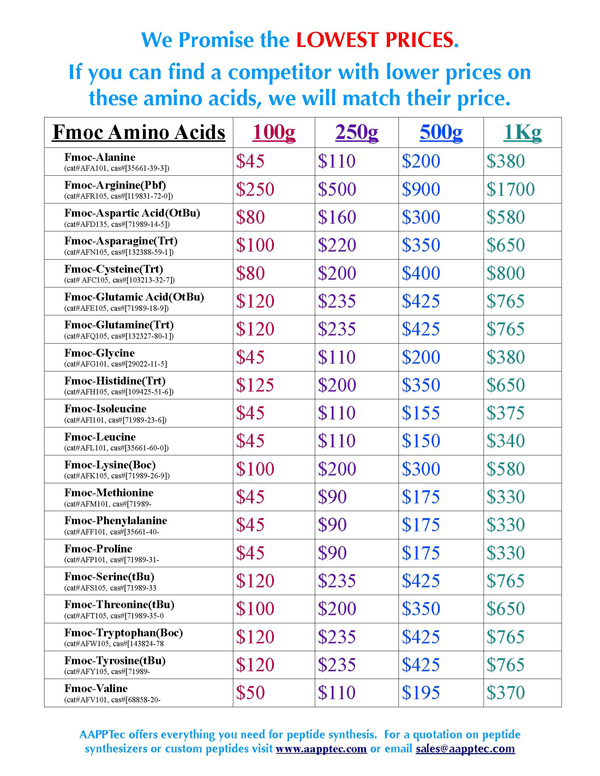
Disomers are also available from AAPPTec. |
|
AAPPTec:
AAPPTec provides high
quality custom peptides
quickly at very competitive
prices. Our peptide chemists
have prepared peptides with up
to 80 to 100 residues in high
purity. They have decades of
experience preparing and
purifying all types of peptides,
including hydrophobic
peptides, cyclized peptides and
peptides conjugated to biotin,
fatty acids or dyes. Most
modifications, such as
phosphorylation, unusual
amino acids, side chain labels
and fluorescent tags, can be
incorporated.
Pseudoproline Dipeptides
Pseudoproline dipeptides
have many uses in peptide
synthesis. They are often
utilized in the synthesis of
difficult, hydrophobic
sequences. The
pseudoproline structure
imposes a bend in the peptide
backbone that disrupts
hydrogen bonding patterns
of the peptide and reduces
the tendency of the peptide
chain to aggregate.
Oxytocin
Oxytocin acts both as a
hormone and a
neurotransmitter in the
brain. The primary effects of
oxytocin are uterine
contraction and facilitating
milk production in lactating
mothers. It is used to induce
labor and toaid milk
production in breast-feeding
mothers. Due to its
structural similarity to
vasopressin, oxytocin can
reduce the production of
urine slightly and in some
species it can stimulate
sodium excretion by the
kidneys. Under certain
circumstances, it can inhibit
the release of
adrenocorticotropic hormone
and cortisol indirectly.
Apex 396 HT Library Synthesizer The Apex 396 HT is unsurpassed for highthroughput synthesis of peptide libraries. It synthesizes up to 192 different sequences at the same time using standard 1 mL well titer plates. The Apex 396 HT is ideal for SAR studies. It can prepare complete alanine scan libraries, deletion libraries and truncation libraries to identify critical segments and residues. The library peptides are cleaved into standard 96 well titer plates.
AAPPTec will be at the American Peptide Symposium in Orlando, Florida this year from June 20-25, 2015! New aspects of the structure and mode of action of the human cathelicidin LL-37 revealed by the intrinsic probe p-cyanophenylananine Xhindoli D, Morgera F, Zinth U, Rizzo R, Pacor S, Tossi A., Biochem. J., 2015, 465, 443-57. doi: 10.1042/BJ20141016 The human cathelicidin peptide LL-37 is an important effector
of our innate immune system and contributes to host defense
with direct antimicrobialactivity and immunomodulatory
properties, and by stimulating wound healing. Its sequence has
evolved to confer specific structural characteristics that strongly
affect these biological activities, and differentiate it from
orthologues of other primate species. In the present paper we
report a detailed study of the folding and self-assembly of
this peptide in comparison with rhesus monkey peptide RL-37,
taking into account the different stages of its trajectory from
bulk solution to contact with, and insertion into, biological
membranes. Phenylalanineresidues in different positions
throughout the native sequences of LL-37 and RL-37 were
systematically replaced with the non-invasive fluorescent and IR
probe p-cyanophenylalanine. Steady-state and time-resolved
fluorescence studies showed that LL- 37, in contrast to RL-37,
forms oligomers with a loose hydrophobic core in physiological
solutions, which persist in the presence of biological membranes.
Fourier transform IR and surface plasmon resonance studies
also indicated different modes of interaction for LL-37 and RL-
37 with anionic and neutral membranes. This correlated with a
distinctly different mode of bacterial membrane
permeabilization, as determined using a flow cytometric method
involving impermeant fluorescent dyes linked to polymers of
defined sizes.
|
 |
UPCOMING EVENTS |
|
May 2015 TIDES May 3-6 May 3-8 Immunology 2015 May 8-12
|
 |


